What Is Yutiq (fluocinolone acetonide)
Yutiq (Fluocinolone ophthalmic (for the eye) implant) is a steroid medicine that is used to treat diabetic macular edema, a build-up of fluid in the retina that is caused by leaking blood vessels.
Yutiq is also used to treat posterior uveitis, inflammation that affects the back of the eye.4
This implant slowly releases fluocinolone into the eye over a period of approximately 30 to 36 months.
Yutiq may also be used for purposes not listed in this medication guide.
An eye implant called Iluvien (fluocinolone) injects medicine into your eyes to treat diabetic macular edema. The fluocinolone implant brands Retisert and Yutiq are used to treat chronic eye inflammation that isn’t brought on by infections (e.g., eye damage, disease). A medical professional must place the implant in your eye, and it can last up to three years.
Uses And Indications For Yutiq
For the treatment of chronic non-infectious uveitis affecting the posterior segment of the eye, YUTIQ (fluocinolone acetonide intravitreal implant) 0.18 mg is recommended.
Yutiq Dosage And Administration
General Dosing Information
For ophthalmic intravitreal injection.
Administration
Sterile gloves, a sterile drape, a sterile caliper, and a sterile eyelid speculum (or equivalent) should all be used during the aseptic intravitreal injection technique. Before the injection, a broad-spectrum microbicide and sufficient anesthetic should be administered.
The injection procedure for YUTIQ is as follows:
- Apply topical and/or subconjunctival anesthetic at the injection site just before injection (inferotemporal quadrant suggested).
- In the lower fornix, apply two to three drops of a broad-spectrum microbicide. Cotton-tipped applicators soaked in a broad-spectrum microbicide can be used to clean the lids. Put a sterile speculum lid in place. Ask the patient to examine the injection site and apply more microbicide solution. Before injecting YUTIQ, give the topical antiseptic 30 to 60 seconds to dry.
- YUTIQ should be positioned posterior to the eye’s equator and inferior to the optic disc. Using callipers, measure 4 millimeters inferotemporal from the limbus to determine the point of entry into the sclera.
- Using sterile procedure, open the sterile foil pouch containing YUTIQ.
- Remove the YUTIQ applicator from the sterile pouch by grasping the barrel of the applicator; do not grasp the plunger.
- Remove the black plunger stop from the plunger.
- Carefully remove the protective cap from the needle and inspect the needle tip to ensure it is not bent.
- Remove the trombone wire from the distal end of the needle. Prior to injection, keep the applicator tip above the horizontal plane to ensure that the YUTIQ implant does not fall out of the applicator.
- Gently displace the conjunctiva so that after withdrawing the needle, the conjunctival and scleral needle entry sites will not align. Care should be taken to avoid contact between the needle and the lid margin or lashes.
- Insert the needle through the conjunctiva and sclera up to the positive stop of the applicator.
- Depress the plunger at the back of the applicator fully to deliver the YUTIQ implant into the back of the eye.
- Remove the YUTIQ applicator from the eye and discard in biohazard sharps container.
- Remove the lid speculum and perform indirect ophthalmoscopy to verify adequate central retinal artery perfusion, absence of any other complications, and to verify the placement of the implant. Scleral depression may enhance visualisation of the implant. Immediate measurement of intraocular pressure (IOP) may be performed at the discretion of the ophthalmologist.
Patients should be watched for endophthalmitis and changes in intraocular pressure after the injection. Tonometry within 30 minutes of the injection, biomicroscopy between two and seven days after the injection, and a check for perfusion of the optic nerve head shortly after the injection are examples of monitoring. Patients should be advised to report any endophthalmitis-related symptoms right away.
Dosage Forms And Strengths
Following the injection, patients should be monitored for changes in intraocular pressure and endophthalmitis. Examples of monitoring include tonometry within 30 minutes of the injection, biomicroscopy between two and seven days after the injection, and a quick check for optic nerve head perfusion. Patients should be instructed to report any symptoms associated with endophthalmitis immediately.
Contraindications
Ocular or Periocular Infections
Patients with active or suspected ocular or periocular infections, such as the majority of viral illnesses of the cornea and conjunctiva, such as active epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, mycobacterial infections, and fungal diseases, should not take YUTIQ.
Hypersensitivity
YUTIQ is contraindicated in patients with known hypersensitivity to any components of this product.
Warnings And Precautions
Intravitreal Injection-related Effects
Endophthalmitis, eye inflammation, elevated or lowered intraocular pressure, and choroidal or retinal detachments have all been linked to intravitreal injections, including those containing YUTIQ. Within 24 hours of the injection, hypotony was noticed, and it went away in two weeks. After the intravitreal injection, patients should be closely watched.
Steroid-related Effects
YUTIQ and other corticosteroids can cause glaucoma, elevated intraocular pressure, and posterior subcapsular cataracts. Corticosteroid usage may promote the development of bacterial, fungal, or viral secondary eye infections.
Patients with a history of ocular herpes simplex should not use corticosteroids due to the possibility of the virus reactivating.
Risk Of Implant Migration
Patients who have a tear or lack the posterior capsule of their lens are susceptible to implant migration into the anterior chamber.
Side Effects And Adverse Reactions
Clinical Studies Experience
Adverse reaction rates found in a drug’s clinical trials cannot be directly compared to those found in another drug’s clinical trials since clinical trials are carried out under very different conditions, and they might not accurately represent rates seen in real-world situations.
Cataract development and subsequent cataract surgery, increased intraocular pressure, which may be linked to optic nerve damage, visual acuity and field defects, secondary ocular infection from pathogens like herpes simplex, and perforation of the globe where there is thinning of the cornea or sclera are all adverse reactions linked to ophthalmic steroids, including YUTIQ.
Patients with non-infectious uveitis affecting the posterior segment of the eye were treated once with either YUTIQ or sham injection in Studies 1 and 2, which were multicenter, randomized, sham injection-controlled, masked trials. After that, they received routine therapy for the remainder of the study. In Study 3, a multicenter, randomized, masked trial, patients with non-infectious uveitis affecting the posterior portion of the eye received routine care for the duration of the study after receiving one treatment with YUTIQ using one of two different applicators.
Data from trials 1, 2, and 3 during a 12-month period for study eyes treated with YUTIQ (n=226) or sham injection (n=94) are compiled in Table 1. Tables 1 and 2 display the most frequent ocular (study eye) and non-ocular adverse responses.
Yutiq Description
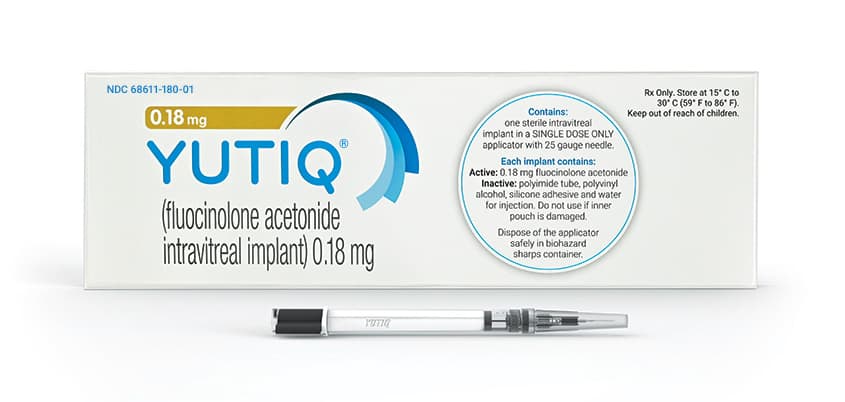
YUTIQ is a sterile, non-bioerodible intravitreal implant with a 36-month sustained-release medication delivery system that contains 0.18 mg of fluocinolone acetonide. The initial rate of fluocinolone acetonide release from YUTIQ is 0.25 mcg/day. To make it easier to inject the implant straight into the vitreous, YUTIQ is preloaded into a single-dose applicator. The medication is fluocinolone acetonide, a synthetic corticosteroid.
Fluocinolone acetonide is known chemically as (6α,11β,16α)-6,9-difluoro-11,21-dihydroxy-16,17-[(1-methylethylidene)bis-(oxy)]-pregna-1,4-diene-3,20-dione.
Fluocinolone acetonide is a white or almost white, microcrystalline powder that is soluble in methanol, ethanol, acetone, and chloroform but very slightly soluble in ether. It is nearly insoluble in water.
A light brown 3.5 x 0.37 mm implant with 0.18 mg of the active substance fluocinolone acetonide and the following inactive ingredients—polyimide tube, polyvinyl alcohol, silicone glue, and water for injection—makes up each YUTIQ.
How Is Yutiq Supplied
YUTIQ (fluocinolone acetonide intravitreal implant) 0.18 mg is supplied in a sterile single-dose preloaded applicator with a 25-gauge needle, packaged in a sealed sterile foil pouch inside a sealed Tyvek pouch inside a carton box.
NDC 68611-180-01
Storage: Store at 15° C to 30° C (59° F to 86° F).