What Is Fluovix Plus (floucinonide)
Fluovix Plus (Fluocinonide) is a strong topical steroid that’s applied on the skin. It treats rashes and skin irritation in adults and children. The medication comes as a cream, ointment, gel, and topical solution. All topical forms come as generics, but the cream is also available under the brand name Vanos. You apply fluocinonide to the affected skin one to four times a day, depending on the formulation. This medication isn’t meant for long-term use because of potential side effects like skin thinning and changes in skin color.
Fluocinonide (Vanos) is a strong topical steroid that treats skin inflammation and irritation, but it’s not meant for long-term use. The medication comes in many different forms, such as a cream, ointment, gel, and topical solution.
How Fluovix Plus (fluocinonide) Works
Fluovix plus (Fluocinonide) is a topical corticosteroid. It works by blocking inflammatory chemicals that cause redness and swelling in the skin. This helps relieve skin irritation and itching due to certain skin problems.
Indications And Usage Of Fluovix Plus
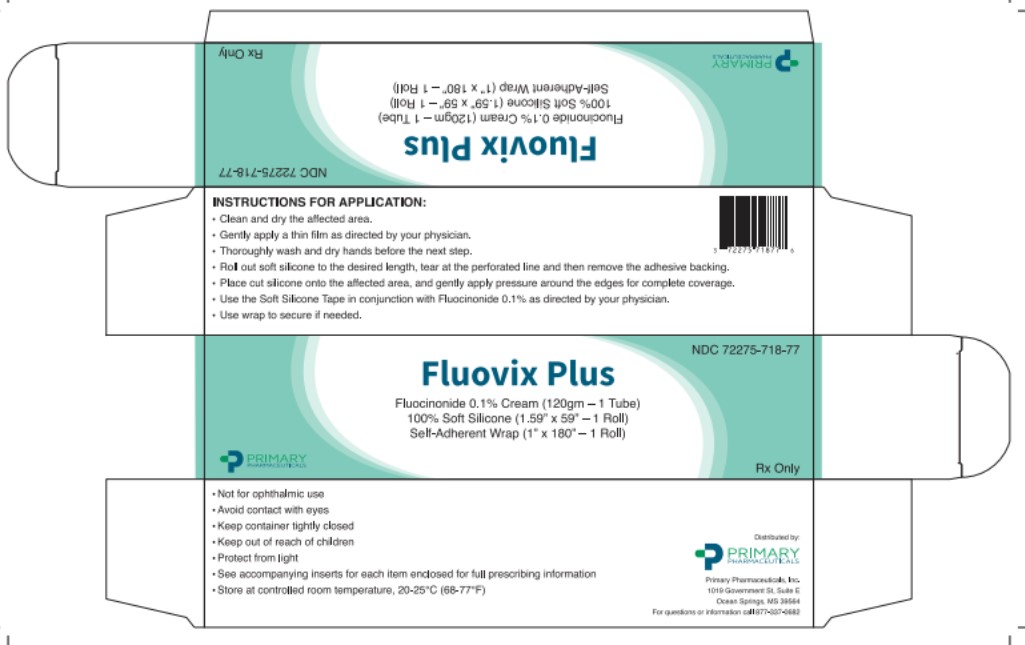
Instructions for Use
- Clean and dry the affected area.
- Gently apply a thin film–1-2 grams, 1-2 times daily or as directed by your physician.
- Do not apply over a large area of skin.
- Thoroughly wash and dry hands before the next step.
- Roll out Soft Silicone to the desired length, tear at the perforated line and then remove adhesive backing.
- Place Soft Silicone onto the affected area, and gently apply pressure around the edges for complete coverage.
- Use the Soft Silicone tape in conjuction with Fluocinonide Cream 0.1% only as directed by your physician.
Significance
For patients 12 years of age or older, Fluovix plus (Fluocinonide Cream USP, 0.1%), is recommended to relieve the inflammatory and itchy symptoms of corticosteroid-responsive dermatoses.
Restrictions On Use
Because the safety of Fluovix plus (Fluocinonide Cream USP, 0.1%) for longer than two weeks has not been established and because the drug may suppress the hypothalamic-pituitary-adrenal (HPA) axis, treatment beyond two consecutive weeks is not advised, and the total dosage should not exceed 60 g per week. Once the disease is under control, therapy should be stopped. Reevaluating the diagnosis can be required if no improvement is observed after two weeks. Use no more than half of the 120 g tube each week.
Fluovix plus (Fluocinonide Cream USP, 0.1%) should not be used to the face, groin, or axillae, nor should it be used to treat perioral dermatitis or rosacea.
Fluovix Plus Cream Dosage And Administration
For topical use only. Fluovix plus (Fluocinonide Cream USP, 0.1%) is not for ophthalmic, oral, or intravaginal use.
As instructed by a doctor, apply a thin coating of Fluovix Plus (Fluocinonide Cream USP, 0.1%) once or twice a day to the skin areas affected by psoriasis. It has been demonstrated that twice-daily application is more successful in treating psoriasis over the course of two weeks.
As instructed by a doctor, apply a thin coating of Fluovix Plus (Fluocinonide Cream USP, 0.1%) once daily to the afflicted skin areas if you have atopic dermatitis. It has been demonstrated that once-daily application is just as successful in treating atopic dermatitis as twice-daily treatment over the course of two weeks.
As instructed by a doctor, apply a thin layer of Fluovix Plus (Fluocinonide Cream USP, 0.1%) once or twice daily to the afflicted regions for corticosteroid-responsive dermatoses other than psoriasis or atopic dermatitis.
Dosage, Forms And Strengths Of Flouvix Plus
Cream, 0.1%.
Each gram of Fluocinonide Cream USP, 0.1% contains 1 mg of fluocinonide in a white to off-white cream base.
Warnings And Precautions Of Fluovix Plus
Effect On Endocrine System
Clinical glucocorticosteroid insufficiency may result from the reversible suppression of the hypothalamic-pituitary-adrenal (HPA) axis caused by systemic absorption of topical corticosteroids, such as Fluocinonide Cream USP, 0.1%. This could happen while the topical corticosteroid is being used or after it is stopped. Furthermore, using Fluocinonide Cream USP, 0.1% for more than two weeks may weaken the immune system.
Fluocinonide cream, 0.1%, applied once or twice daily has been shown to suppress the HPA axis in two out of eighteen adult patients with plaque-type psoriasis, one out of thirty-one adult patients with atopic dermatitis, and four out of 123 pediatric patients with atopic dermatitis.
Patients using topical corticosteroids, such as Fluocinonide Cream USP, 0.1%, may need to have their HPA axis suppression assessed on a regular basis due to the possibility of systemic absorption. A patient using a topical corticosteroid may be more susceptible to HPA axis suppression if they use stronger steroids, apply them to large regions of skin, use them for extended periods of time, use them under occlusion, use them on altered skin barriers, or use them in patients with liver failure.
When assessing patients for HPA axis suppression, an ACTH stimulation test may be useful. If there is evidence of HPA axis suppression, the medication should be progressively stopped, the frequency of administration should be decreased, or a less strong steroid should be used in its place. Adrenal insufficiency symptoms may necessitate further systemic corticosteroids. When topical corticosteroids are stopped, HPA axis function usually recovers quickly and completely.
Systemic absorption of topical corticosteroids can potentially cause Cushing’s syndrome, hyperglycemia, and latent diabetes mellitus.
The overall systemic absorption of topical corticosteroids may rise if multiple corticosteroid-containing products are used concurrently.
Local Adverse Reactions With Topical Corticosteroids
Higher potency corticosteroids, extended use, or occlusive use may increase the risk of local adverse effects. Atrophy, striae, telangiectasis, burning, itching, irritation, dryness, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria are examples of reactions. Certain negative local effects might not be reversible.
Concomitant Skin Infections
An appropriate antifungal or antibacterial medication should be given if concurrent skin infections are present or develop. Use of Fluocinonide Cream USP, 0.1% should be stopped until the infection has been sufficiently managed if a positive reaction does not happen quickly.
Allergic Contact Dermatitis
Fluocinonide Cream USP, 0.1% should be stopped and the proper treatment should be started if irritation appears. Unlike most topical treatments that do not contain corticosteroids, allergic contact dermatitis with corticosteroids is typically diagnosed by observing failure to heal rather than documenting a clinical aggravation. Appropriate diagnostic patch testing should be used to confirm such an observation.
Adverse Reactions/Side Effects
As your body adjusts to the drug, the following adverse effects might become less severe over time. If these symptoms persist or get worse over time, let your doctor know right once.
Common Side Effects
- Burning Feeling Where Drug is Applied
Less Common Side Effects
- Dry skin
- Itchy skin
- Irritated skin
- Inflamed hair follicles
- Headache (0.1% cream only)
The following Fluocinonide side effects have also been reported
A medication may have some undesirable side effects in addition to its necessary ones. Even though not all of these adverse effects are possible, they might require medical care if they do.
Incidence not known
- Blistering, burning, crusting, dryness, or flaking of the skin
- Irritation
- Itching, scaling, severe redness, soreness, or swelling of the skin
- Redness and scaling around the mouth
- Thinning of the skin with easy bruising, especially when used on the face or where the skin folds together (e.g. between the fingers)
- Thinning, weakness, or wasting away of the skin
There may be certain adverse effects that typically don’t require medical treatment. As your body becomes used to the medication, these adverse effects can disappear. Additionally, you might be able to learn how to lessen or avoid some of these adverse effects from your health care provider. If any of the following side effects persist, are bothersome, or raise any questions, speak with your healthcare provider:
Less Common
- Fever
- Headache
- Muscle aches
- Sore throat
- Stuffy or runny nose
- Unusual tiredness or weakness
Incidence Not Known
- Acne or pimples
- Burning and itching of the skin with pinhead-sized red blisters
- Burning, itching, and pain in hairy areas, or pus at the root of the hair
- Increased hair growth on the forehead, back, arms, and legs
- Lightening of normal skin color
- Lightening of treated areas of dark skin
- Reddish purple lines on the arms, face, legs, trunk, or groin
- Softening of the skin
Some patients may experience additional adverse effects not on this list. Consult your healthcare provider if you observe any further consequences.